Details
|
Product Name |
Specimen |
Format |
Catalog No. |
Cut-off(ng/ml) |
Strip width |
| (AFP)Alpha-Fetoprotein Rapid Test |
Whole Blood/Serum/plasma |
Strip |
AFP-W01B |
20 |
3.0mm |
|
Strip |
AFP-W01D |
20 |
4.0mm |
INTRODUCTION
One Step strip Style AFP Test in whole blood is a rapid direct binding test for the detection of Alpha Fetoprotein in whole blood as an aid in the diagnosis of primary hepatocellular carcinomas, testicular teratocarcinomas and neural tube defects. The test is based on the principle of sandwich immunoassay for determination of AFP. Monoclonal and polyclonal antibodies are employed to identity AFP specifically. This one step test is very sensitive and only takes about 10-20 minutes. The sensitivity of the test can reach to 20ng/mL.
SPECIMEN COLLECTION
For whole blood, collect blood into a container with anticoagulant. If the specimen cannot be tested on the day of collection, store the whole blood specimen in a refrigerator. Bring the specimens to room temperature before testing.
TEST PROCEDURE
1. When you are ready to begin testing, open the sealed pouch by tearing along the notch. Remove the test from the pouch and use it as soon as possible. Best results will be obtained if the assay is performed within one hour.
2. Place the test device on a clean and level surface. Be sure to label the device with specimen’s ID number.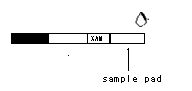
3. lay the strip flat on a clean, dry, nonabsorbent surface ,draw one drop (about 20ul)sample into the sample pad.
4. Then draw one drop of buffer into the sample pad as well and start the timer.
5. Wait fot the red line(s) to appear. The result should be read at 10-15 minutes. Do not read results after 20 minutes.
INTERPRETATION OF RESULTS
Negative:Only one colored band appears on the control (C) region. No apparent band on the test (T) region.
Positive:In addition to a pink colored control (C) band, a distinct pink colored band will also appear in the test (T) region. This indicates an AFP concentration of more than 20ng/mL.If the test band is equal to or darker than the control band, it indicates that the AFP concentration of specimen has reached to or is greater than 400ng/mL. Please consult your physician to perform a much more detailed exam.
Invalid:A total absence of color in both regions is an indication of procedure error and/or that the test reagent has deteriorated.
STORAGE AND STABILITY
The test kits can be stored at room temperature (2 to 30C) in the sealed pouch to the date of expiration. The test kits should be kept away from direct sunlight, moisture and heat.
PRECAUTION
1. For in vitro diagnostic use only.
2. Do not use test kit beyond the expiry date.
3. The test device should not be reused.




-Carcinoembryonic-Antigen-Test-Strip-CEA-W01B.96.1.jpg)