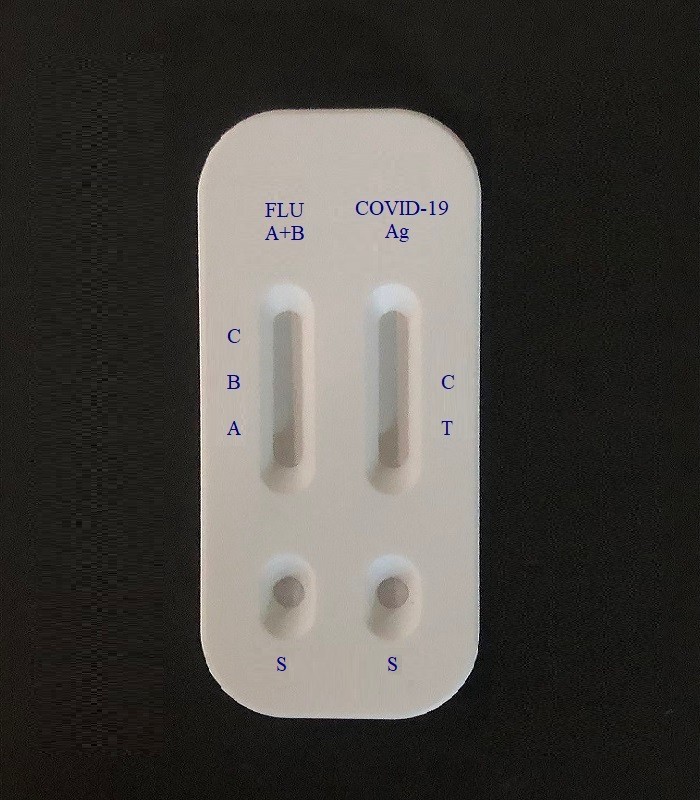
Intended use
The Influenza A+B & COVID-19 (SARS-CoV-2) Antigen Test Kit is an in vitro immunochromatographic assay for the qualitative and differential detection of nucleocapsid protein antigen from influenza A, influenza B and SARS-CoV-2 in pharyngeal swab and nasal swab specimens. It is intended to aid in the rapid diagnosis of influenza A, influenza B and SARS-CoV-2 infections.
Influenza, usually called "flu", is an acute respiratory infectious disease caused by influenza viruses. It is highly contagious and is spread mainly through coughing and sneezing. It usually breaks out in spring and winter. Divided into influenza A virus, influenza B virus and influenza C virus. Influenza A virus has strong variability, followed by influenza B virus, and influenza C virus is very stable, so influenza A virus is more serious and prevalent than influenza B virus. The novel coronaviruses belong to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection, asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main Manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
Material Provided
1.Test Devices
2.Extraction Buffer
3.Extraction Tube
4.Sterilized Swab
5.Work Station
6.Package Insert
Specimen Collection
1. Oropharyngeal Swab Sample:
Let the patient's head tilt slightly, mouth open, and make "ah" sounds, exposing the pharyngeal tonsils on both sides. Hold the swab and gently wipe both sides of the patient's pharyngeal tonsils back and forth at least three times.
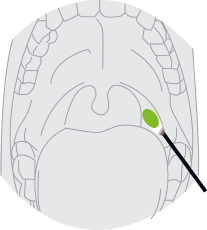
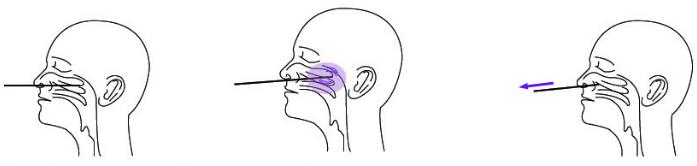
2. Nasopharyngeal Swab Sample:
Let the patient's head relax naturally, carefully insert the swab in the patient’s nostril. Swab over the surface of the posterior nasopharynx and rotate the swab several times.Use the same swab, take specimens from the other nostril in the same way.
After Swab specimens were collected, swab can be stored in extraction reagent provided with the kit. Also can be stored by immersing the swab head in a tube containing 2 to 3 mL of virus preservation solution (or isotonic saline solution, tissue culture solution, or phosphate buffer).
Freshly collected specimens should be processed as soon as possible, but no later than one hour after specimen collection. Specimen collected may be stored at 2-8℃ for no more than 24 hours; Store at -70 ℃ for a long time, but avoid repeated freeze-thaw cycles.
Specimen Preparation
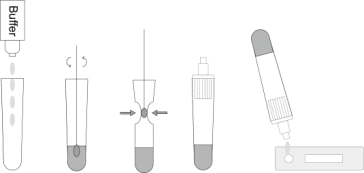
1. Take out the extraction tube, add 8 drops (about 0.3 mL) of the extraction reagent into the extraction tube, and put it on the tube rack.
2. Put the swab specimen into the extraction tube, rotate the swab for about 10 seconds, and press the swab head against the tube wall to release the antigen in the swab.
3. Remove the swab while squeezing the sides of the tube to extract the liquid from the swab. so as to remove as much liquid as possible from the swab. Dispose of swabs according to biohazard waste disposal method.
4. Insert a dropper tip into the extraction tube tightly.
Read the instructions carefully before use and allow test device, extraction reagent and specimens to equilibrate to room temperature prior to testing.
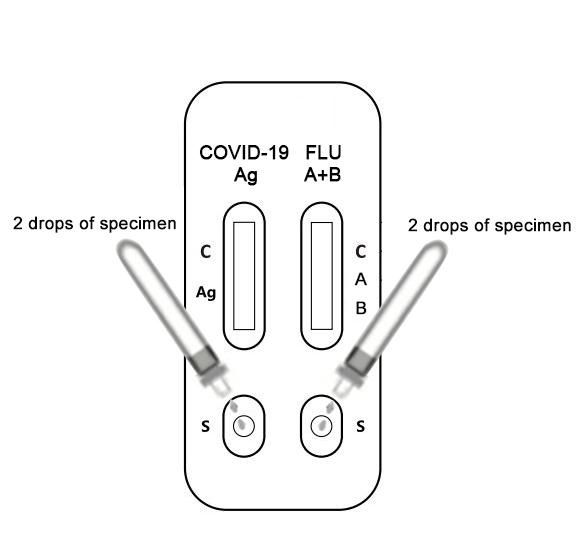
1. Open the package and take out the test device.
2. Hold the extraction tube vertically and add two drops of the test specimens into the specimen well (s). Start the timer.
3. Interpret the results within 20 minutes. Strong positive results can be reported within 20 minutes, however, negative results must be read after 20 minutes, and the results after 30 minutes are no longer valid.
Interpretation of Test Results
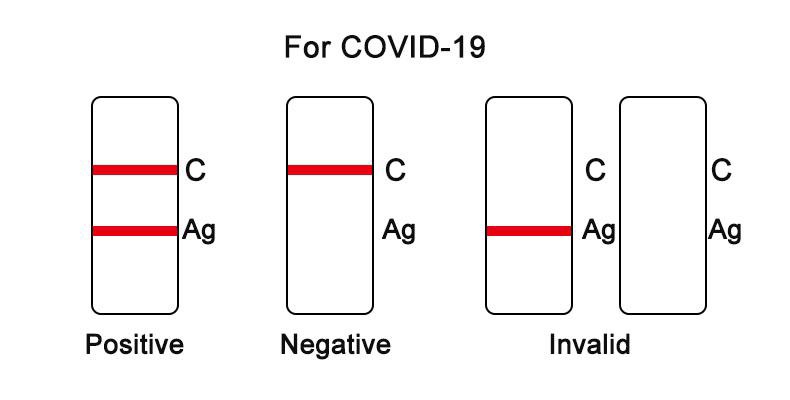
FOR COVID-19 ANTIGEN:
Negative result: if there is only a quality control line C, the detection line Ag is colorless, indicating that SARS-CoV-2 antigen has not been detected and the result is negative.
Positive result: if both the quality control line C and the detection line Ag appear,indicating that SARS-CoV-2 antigen has been detected and the result is positive.
Invalid result: if the quality control line C is not observed, it will be invalid regardless of whether there is detection line Ag (as shown in the figure below), and the test shall be conducted again.
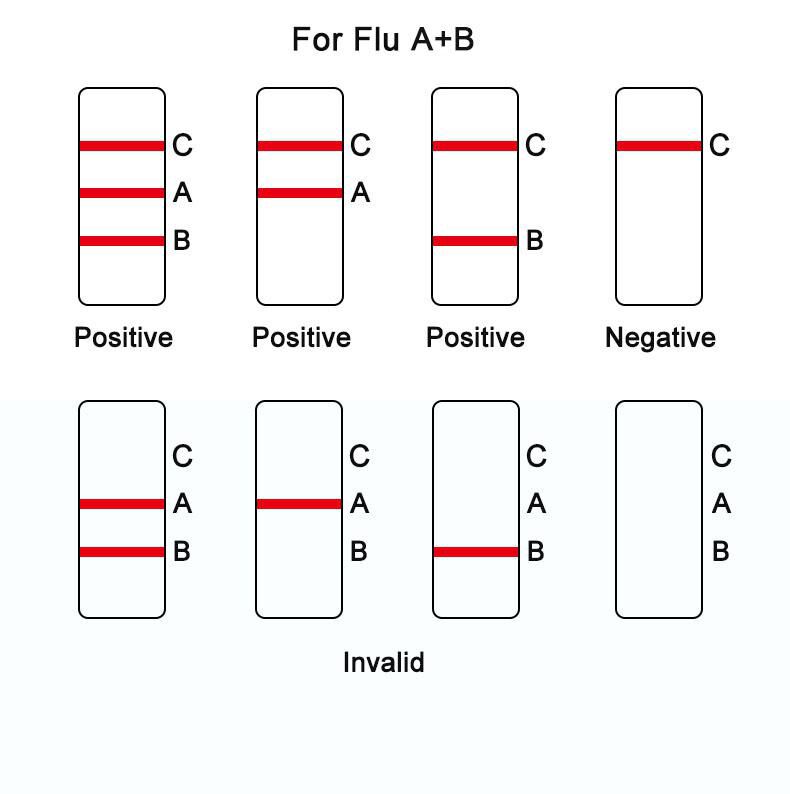
FOR INFLUENZA A+B ANTIGEN:
Negative result: if there is only a quality control line C, the detection lines A&B are colorless, indicating that both influenza A&B antigen have not been detected and the result is negative.
Positive result:1) if the quality control line C and the detection line A&B all appear, indicating that influenza A&B antigens has been detected and the result is positive;
2) if both the quality control line C and the detection line A appear, indicating that influenza A antigen has been detected and the result is positive;
3) if both the quality control line C and the detection line B appear, indicating that influenza B antigen has been detected and the result is positive;
Invalid result: if the quality control line C is not observed, it will be invalid regardless of whether there is detection