INTENDED USE
This test is a single use, rapid device intended for qualitative detection of antibodies to Human Immunodeficiency Virus 1/2 in Saliva. It is intended for use in medical institution as an aid for the diagnosis and management of patients related to infection with HIV .
SUMMARY
HIV is the etiologic agent of Acquired Immune Deficiency Syndrome (AIDS). The virus is surrounded by a lipid envelope that is derived from host cell membrane. Several viral glycoproteins are on the envelope. Each virus contains two copies of positive-sense genomic RNAs. Patients with HIV-1 has been isolated from patients with AIDS and AIDS-related complex and from healthy individuals with a high potential risk of developing AIDS. Patients with HIV-2 has been isolated from West Africa AIDS patients and from seropositive asymptomatic individuals. Both HIV-1 and HIV–2 elicit an immune response. Detection of HIV antibodies in whole blood, serum or plasma is the most efficient and common way to determine whether an individual has been exposed to HIV .research shows people living with HIV in addition to the blood samples, urine, saliva, tears, semen and vaginal secretions can detect HIV antibodies.People in oral mucosal exudate can detect HIV 1/2 antibody, rapid, noninvasive, high sensitivity, strong specificity, is easy to be accepted by the tester.
PRINCIPLE
HIV 1+2 test contains a membrane strip, which is pre-coated with recombinant HIV 1 capture antigen (gp41) and HIV 2 capture antigen (gp36) in test line . The anti human IgG- colloidal gold conjugate and saliva specimen move along the membrane chromatographically to the test region (T), forming a visible line as the antigen-antibody-anti human IgG particle complex forms with high degree of sensitivity and specificity. This test device has letters of T and C as “Test Line", and "Control Line" on the surface of the cassette. The Test Line and Control Line in result window are not visible before applying any specimen. The Control Line is used for procedural control. Control line should always appear if the test procedure is performed properly and the test reagents of control line are working.
MATERIALS PROVIDE
HIV 1+2 Test Device (Saliva) contains following items to perform the assay:
1. HIV 1+2 test device
2. Instruction for use
3. Chasing buffer
4.Oral swabs
5. Pipette
PRECAUTIONS
1. Read instruction for use carefully before performing this test.
2. For in vitro diagnostic use only.
3. Do not use the test device beyond the expiration date.
4. The test device should remain in the sealed pouch until use. Do not use the test device if the pouch is damaged or the seal is broken.
5. Do not reuse the device.
6. Treat and properly handle the specimens and used device as if they were potentially infectious.
STORAGE AND STABILITY
The test device should be stored at 2-30℃ in the sealed pouch. Avoid humidity, heat and direct sunlight. The test device is stable through the expiration date printed on the sealed pouch. DO NOT FREEZE.
WARNINGS
1. There should be no eating, drinking or smoking where specimens are being handled.
2. Wear disposable gloves and lab coat while handling specimens. Wash hands thoroughly afterwards.
3. Avoid splashing or aerosol formation.
4. Clean spills thoroughly using an appropriate disinfectant.
SPECIMEN COLLECTION AND STORAGE
Read instruction for use carefully before performing this test. In accordance with the provisions of the sampling type, careful operation points and pollution prevention measures, to ensure the accuracy of test results.
Specimen collection
1.Gargle with warm water before collection.
2.Remove one of the clean swabs provided from the bag. Grasp the swab by the handle. Avoid touching the cloth end of the swab.
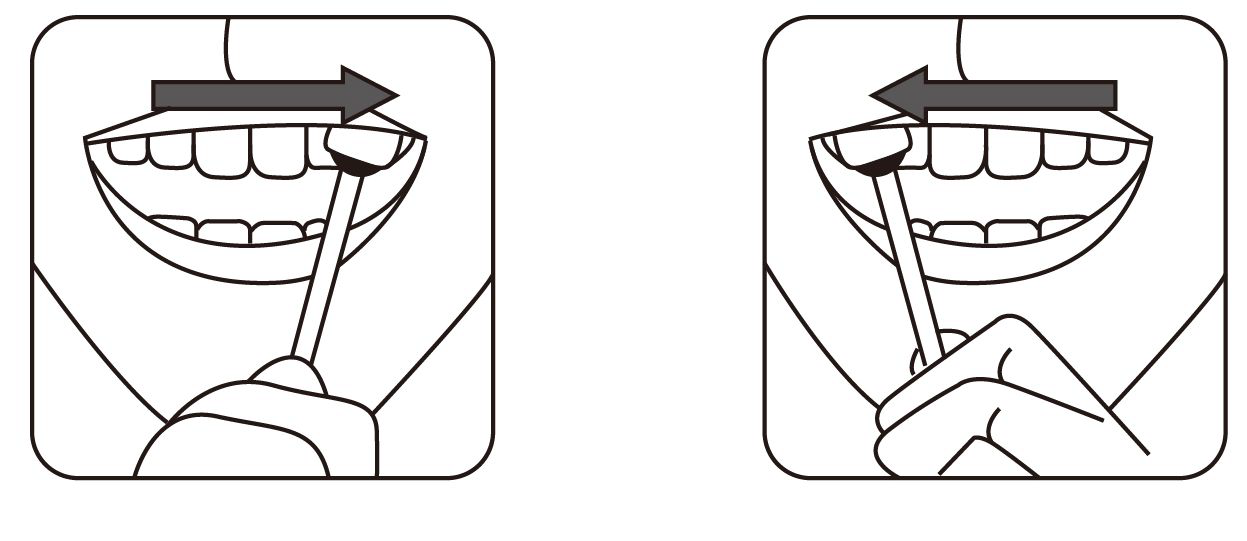
3.Apply moderate pressure while gently swabbing the upper gum line back and forth with the cloth end of the swab. Begin at one corner of the mouth, swabbing gently and slowly until reaching the other corner of the mouth and then swab back across the upper gum line to where you started (about 5-6 seconds).
4. Turn the swab to use the other side of the swab for the lower gums.
5.Using the other side of the swab, gently and slowly swab the lower gum line back and forth. Begin at one corner of the mouth, ending at the other corner of the mouth and then swab back across the lower gum line to where you started(about 5-6 seconds).
Note: Procedure collection step strictly according to requirements. The specimen shall be the oral mucosal exudate not saliva.
Specimen Treatment
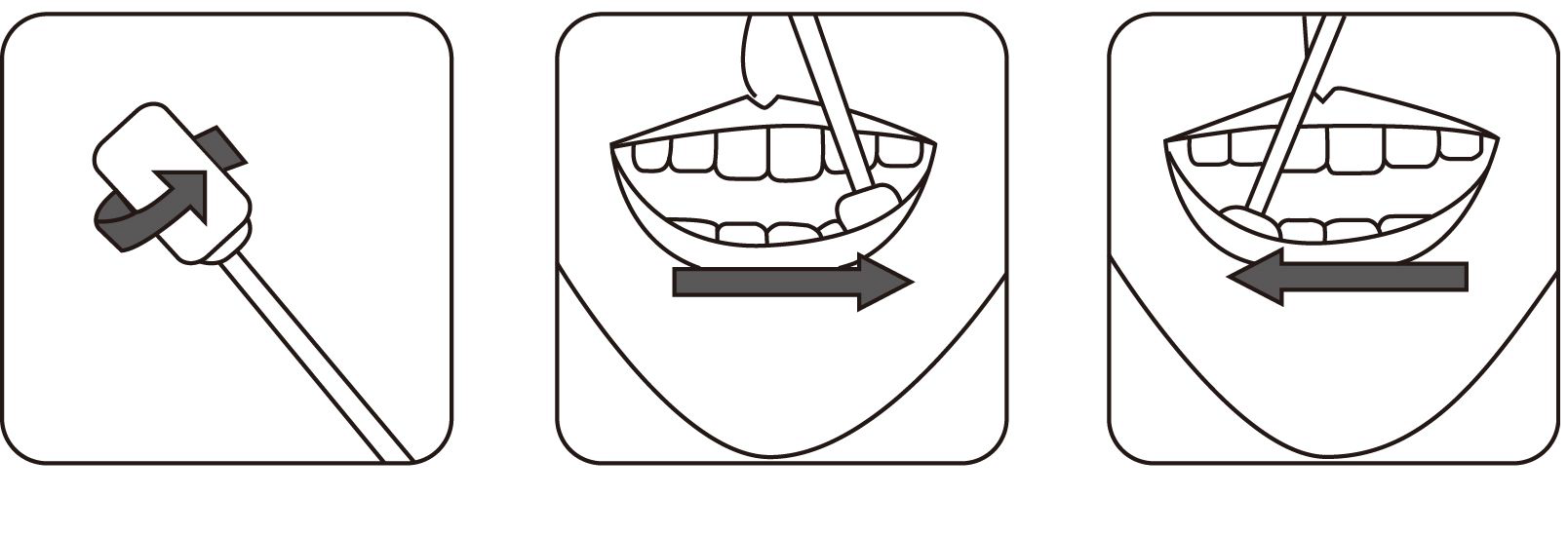
1.Immediately place the swab in the tube containing the developing solution.
2.Grasp the swab handle firmly. Plunge the swab up and down 6-8 times in the sample buffer tube, rubbing both sides of the swab against the wall of the tube. Take out the Swab,and the treated solution shall be ready for test.
Note:
1.Specimen shall be treated with in 1 hour after collection at room temperature. If the specimen is not tested immediately, refrigerate it at 2-8℃ and finish the treatment within 12 hours.
2.Do not freeze and thaw the specimen repeatedly.
TEST PROCEDURE
1. Bring the pouched test device to room temperature (15-30℃) prior to testing. Do not open the pouch until ready to perform the assay.
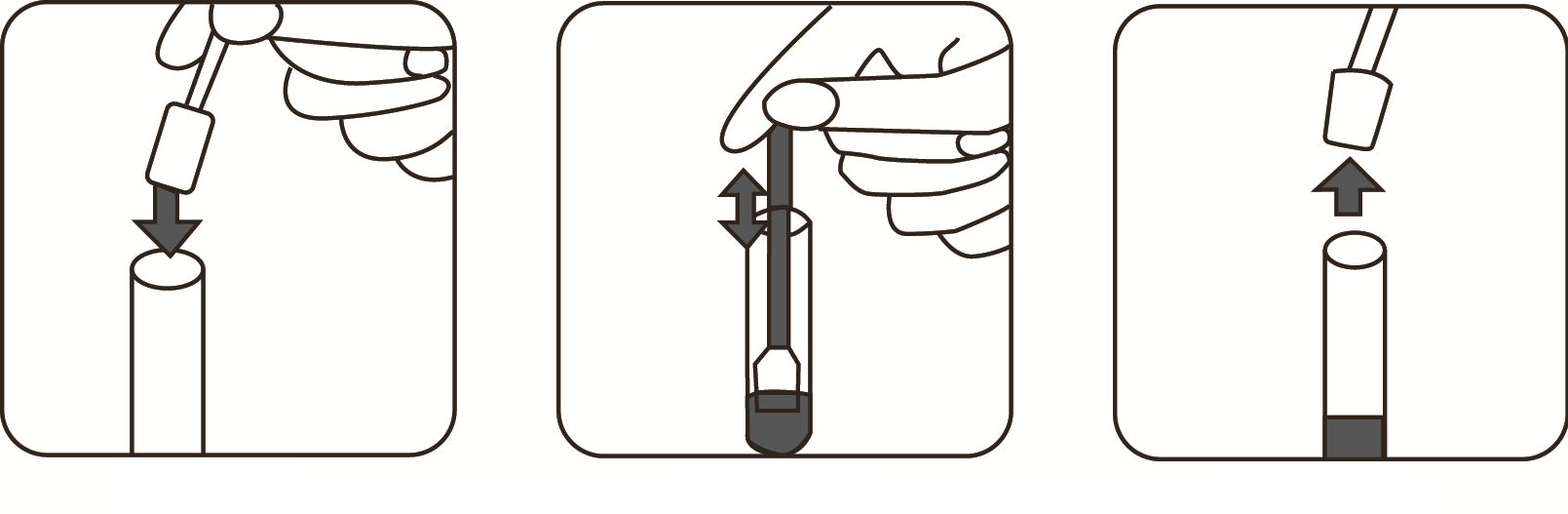
2. Remove the test device from the sealed pouch. Lay it on a flat, clean and dry surface.
3. Use the pipette to draw and slowly add 2 drop of Solution treated sample to the sample well.
4. Read test results between 15-20 minutes. Do not interpret after 20 minutes.
INTERPRETATION OF RESULTS
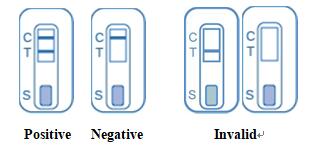
positive for HIV-1 /2 .
Negative: Only the control line appears in the result window. The test result is negative and no antibodies to HIV-1/2 have been detected.
Invalid: If the control line does not appear in the result window, the test results are invalid regardless of the presence or absence of the test line.
NOTE: Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for the control line failure. Review the procedure and repeat the test with a new device. If problem persists, please contact your
local distributor.


2. High sensitive
3. High Accuracy
4. OEM available




